ACTIVE-CLC: Active cardiac locomotor coupling
The ACTIVE project investigates the relationship and the interaction between cardiovascular and locomotor system during normal daily activities and in laboratory. Starting from this knowledge, we aim to explore the beneficial effects of home-based cardiac rehabilitation treatment for patients with heart failure using a wearable sensor system with an auditory signal for the synchronization of the heart and movement.
Publications
A. Rosato et al. (2025) — Frontiers in Network Physiology, vol. 5
A. Rosato, E. Rullman, S.A. Dual (2025) — Computing in Cardiology, vol.52
A. Rosato, S.A. Dual (2024) — Computing in Cardiology (CinC)
A. Rosato et al. (2024) — Frontiers in Physiology, 15
A. Rosato et al. (2023) — BIOSTEC 2023, vol 4
Age inclusive design of interactive wearable for real-time biofeedback
Older adults represent a heterogeneous group regarding their physical health, exercise habits and interests for health monitoring for physical exercise. Wearable devices are often used for short-term purposes, and the long-term engagement with applications remains a challenge for all ages. Despite that technology available, interactive features such as biofeedback have been rarely implemented. There is a need to design age-inclusive interactive wearable technology that take into consideration older adults’ heterogeneity regarding their physical health, digital skills and individual motivation for physical exercise. This project aims to co-design inclusive interactive wearable technologies for enhancing physical activity among older adults to improve the long-term user engagement with wearable technologies over time.
Publications
T.-Y. Lu et al. (2025) — JMIR Preprints

Cardiac support technologies
Aortic Assist
The AorticAssist project investigates cardiovascular autoregulatory mechanisms, soft robotic actuator design, and the clinical use of aortic counterpulsation. This research aims to improve the candidacy of counterpulsation as a long-term, out-of-hospital treatment option for heart failure patients and increase the efficacy of counterpulsation as a method of cardiac recovery. AorticAssist relies on benchtop methods such as mock flow loops, sensorised physiological phantoms, and 4D Flow MRI, as well as in-vivo and clinical studies to improve our understanding of the interaction between circulatory support and cardiovascular autoregulatory responses. These results are then used for data-driven design of novel soft-robotic counterpulsation actuators.
Electrically-driven soft robotic blood flow support to the lungs
Post-operative failure of the right side of the heart is a major complication in cardiac surgery. Inspired by my PostDoc project on extra-vascular compression device, we developed a similar counter-pulsation device for augmenting blood flow to the lungs. A critical extension was the electro-pneumatic actuation approach which holds the potential for transcutaneous power transmission, reducing current electric driveline complications.
Publications
I. Pirozzi, A. Kight, A. Kyungwon Han, R. Cutkosky, S.A. Dual (2022) — Advanced Materials, adma.202210713
I. Pirozzi, A. Kight, X. Liang, A. Kyungwon Han, D.B. Ennis, W. Hiesinger, S.A. Dual, and M. Cutkosky (2022) — Advanced Materials Technologies, 2201196
Soft robotic cardiac assist device for the forgotten halt of heart failure patients
No effective treatment for heart failure with preserved ejection fraction (HFpEF) is available at present. HFpEF is characterized by a pathological reduction in heart compliance, which impairs the ability of the heart to fill properly and results in low cardiac efficacy. I developed a soft robotic cardiac assist device, which wraps around the aorta and reduces the pressures seen by the heart in a counter-pulsation way. Soft robotic actuators allow the device to smoothly conform to the native vasculature and will enable the miniaturization of the device for minimally invasive implantation. In the first step, the device is tested in vitro to prove pressure reduction and effects on flow fields using imaging. In the second step, an in-vivo study in pigs determines if the cardiac output of a living heart could be increased through the soft robotic cardiac assist device.
Publications
S. A. Dual et al. (2023) — European Society of Artificial Organs (ESAO)
S.A. Dual et al. (2022) — ISMRM (Rapid-Fire)
M. Arduini et al. (2022) — Frontiers in Cardiovascular Medicine
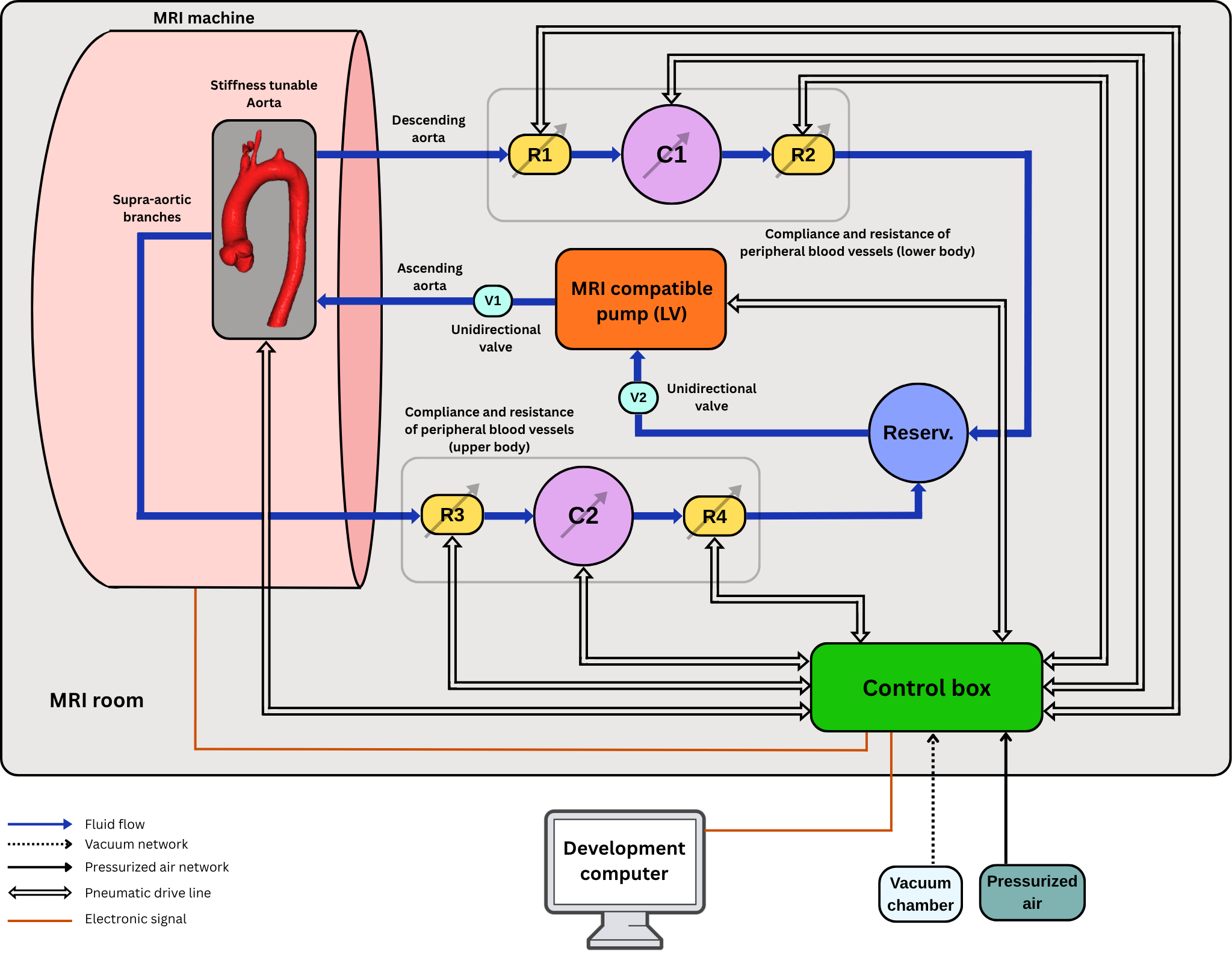
Test-rigs for patient-specific insight
In recent years, biomimetic design from the field of soft robotics play an increasing role in mimicking physiological phenomena on the benchtop. From an ethical perspective, benchtop and computational testing mitigates the need for excessive animal testing, a common practice in medical device innovations. Our research focuses on enabling patient-specific testing with the goal to include underrepresented patient groups such as women and children early in the design process. As a part of this work, we have built Scandinavia's first hybrid mock circulatory loop, which enables testing of physical prototypes in interaction with a virtual model of the patient.
CardioLoop: A versatile hybrid mock circulatory loop for medical device testing
“I feel satisfied as this grant recognises our work in animal-free testing of mechanical assist devices and artificial hearts,” Dual says. In the project, the researchers will develop test rigs that allow for patient-specific testing in combination with magnetic resonance imaging of 4-dimensional flow fields. In contrast to other systems, Dual explains, they can test medical devices in dynamic interaction with the human cardiovascular system. “This allows us to simulate not only average conditions but also tune to patient-specific parameters and simulate sleep or exercise. In this way, we become a realistic alternative to animal trials.”
Patient-specific computational modeling
As cardiovascular diseases continue to be the leading cause of mortality worldwide, computational and hybrid testing of treatment strategies such as heart pumps present a promising avenue for saving lives. However, these models are most often generic and lack patient-specific insight.
Pre-clinical evaluation of the physiological control of a total artificial heart
The focus of this research is to advance the knowledge of total artificial hearts as a viable therapeutic option for patients suffering from heart failure. Using a hybrid mock circulatory loop, we can generate real-time physiological hemodynamic conditions. This dynamic testing platform allows us to simulate various patient states, including exercise conditions, offering valuable insights for improving the design of artificial hearts and other cardiovascular assist devices.
Publications
E. Perra, D. Jonasson, S. Faisal Zaman, T. Finocchiaro, I. L. Perkins, S.A. Dual (2024) — International Society of Mechanical Circulatory Support, Japan
Data-driven cardiovascular assist devices
This research aims at designing and optimizing an algorithm for tuning the parameters of a cardiovascular model to generate patient-specific simulations. The goal is to enhance the accuracy and performance of cardiovascular models, ultimately facilitating the creation of patient-specific digital twins.
Publications
E. Perra, G. Uribarri, E. Fransen, S.A. Dual (2024) — European Society of Biomechanics, Edinburgh
E. Perra, O. Kreis, S.A. Dual (2023) — FIMH 2023, Springer, Cham.
Fundamental understanding of aortic coarctation biomechanics during exercise
The aim of this study was to predict blood pressure gradient (∆P) across the Coarctation of the aorta (CoA) during exercise through in vitro measurements acquired in compliant aortic phantoms incorporated in a hybrid mock circulatory loop tuned to patient-specific medical record data.
Publications
P. J. Nair et al. (2025) — Journal of Biomechanical Engineering, vol. 147, no. 7
P. Nair, E. Perra, D.B. McElhinney, A.L. Marsden, D.B. Ennis, S.A. Dual (2024) — European Society of Biomechanics, Edinburgh (Nominated for Poster Award)
P. J. Nair et al. (2023) — FIMH 2023, pp. 515-523



Physiology-guided signal processing
Digital Health Tools for non-invasive diagnosis of lung disease
Pulmonary hypertension (PH) is a strong prognostic factor in many cardiovascular diseases, such as heart failure, valvular disease and congenital heart disease. Driven by the need for reliable non-invasive techniques, engineering tools must be developed to monitor, detect and predict PH using algorithms which integrate standard echocardiographic images of tricuspid regurgitant and pulmonary flow. Artificial intelligence methods can automate such image analyses, improving robustness.
Publications
P. Sattar, C. Verdonk, F. Hermansson, X. Tang, A. Marsden, F. Haddad, S.A. Dual (2024) — Computing in Cardiology (CinC), pp. 1-4
S.A. Dual, C. Verdonk, M. Amsallem, J. Pham, C. Obasohan, P. Nataf, D.B. McElHinney, T. Kuznetsova, R. Zamanian, J. Feinstein, A. Marsden, F. Haddad (2022) — Pulmonary Circulation, 12:e12125
F.C. Nordgren Hermansson et al.

Implantable and wearable sensors for health monitoring
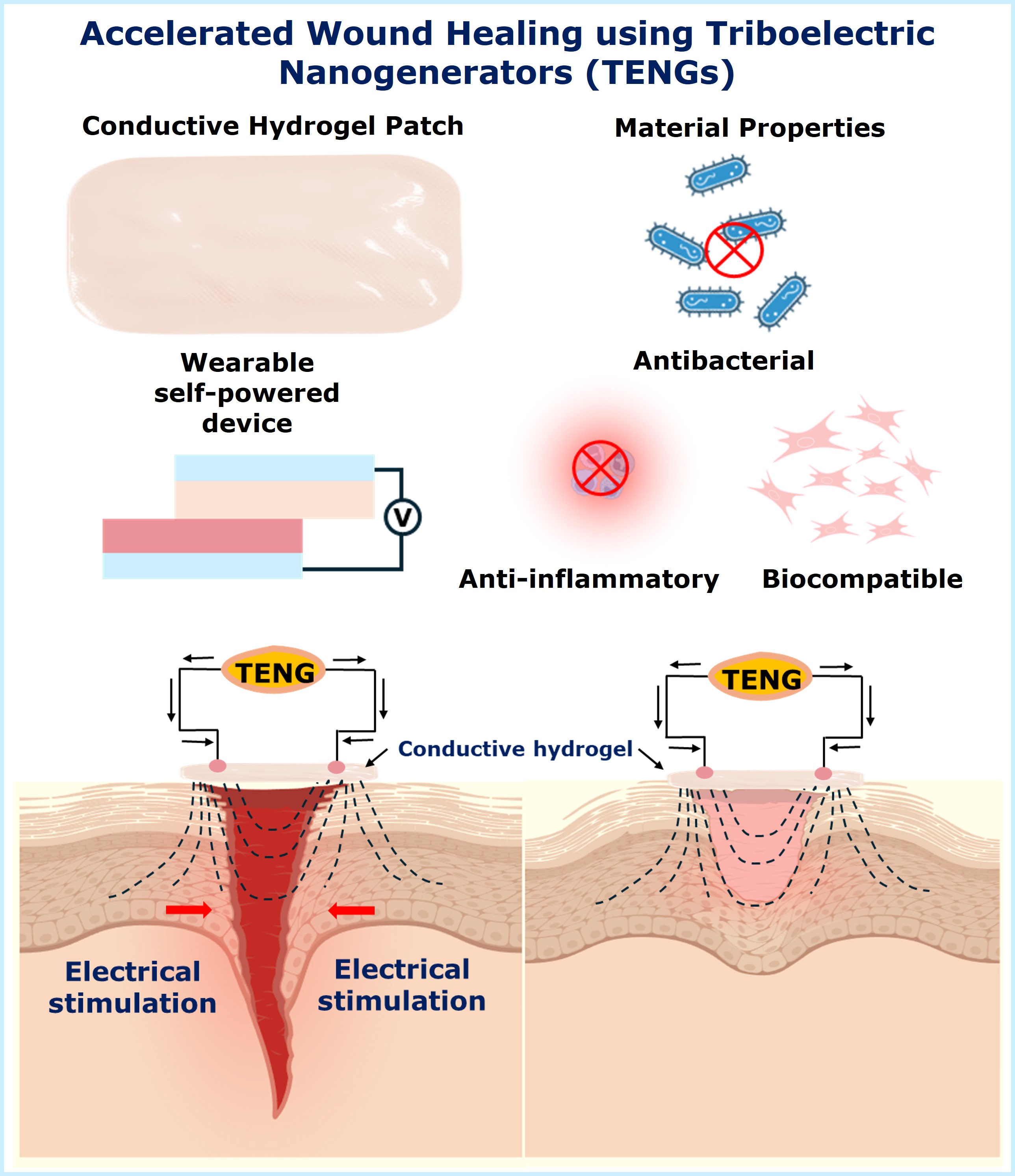
Biocompatible and biodegradable materials based self-powered wearable sensors for healthcare monitoring
Wearable healthcare monitoring systems can monitor vital health parameters such as heart rate, respiration, and movement which can be related with various diseases. Triboelectric nanogenerators (TENGs) are devices which can convert mechanical energy into electrical energy without the need of batteries. TENG based healthcare sensors offer advantages including continuous operation, minimal device complexity, improved biocompatibility, and great wearability.
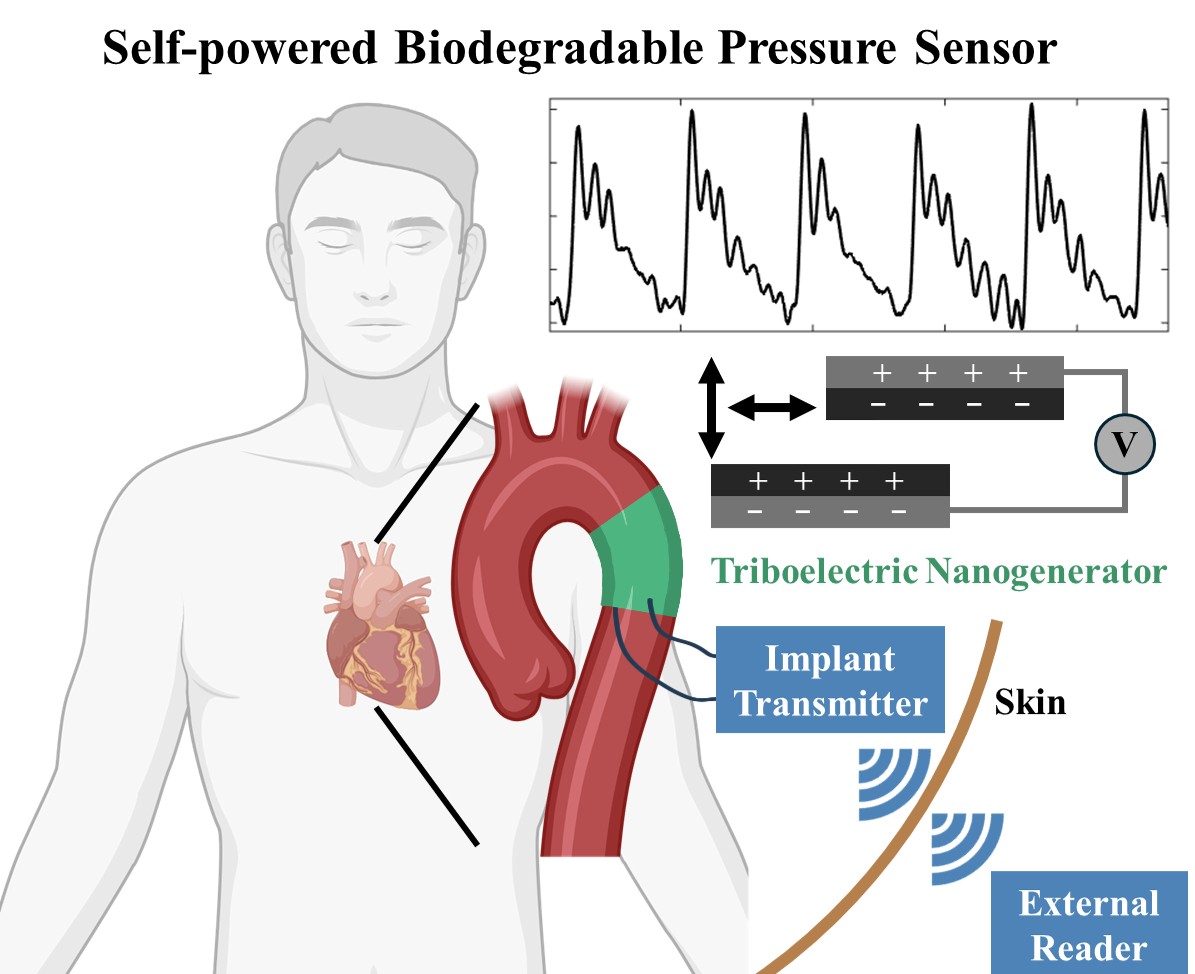
Self-powered biodegradable sensor for post-surgical monitoring
Digital sensors enabling continuous monitoring of blood pressure would allow early discovery of blood vessel collapse or clot – primary causes of readmission after surgery. Our goal is to develop a self-powered biodegradable pressure sensor with the potential for wireless signal transfer that eliminates the need for device-extraction surgeries.
Highly durable soft strain sensor
Building on previous work introducing a durable, soft strain sensor, this research presents VITALS, an implantable strain sensing network that enables continuous biventricular, multiaxial monitoring of cardiac function. Animal studies demonstrate VITALS’ ability to track epicardial strain with high fidelity and detect preload changes rapidly.
Publications
A. Kight, M. Haidar, M. Shibata, Y. Ono, G. Ikeda, A. Sharir, F. Semproni, Y. Palagani, S. Taheri, A. Kyungwon Han, M. Ma, K. Riemer, D.B. McElhinney, S.A. Dual, M. Cutkosky — npj Biomed. Innov. (In Press)
A. Kight et al. (2023) — Sensors, vol. 23, no. 4
A. Kight, M. Cutkosky, D.M. McElHinney, I. Pirozzi, X. Liang, S. Dual, K.W. Han (2023) — Invention disclosure, USPTO 63/443421